





(The example below illustrates the reaction between iodomethane and ethylamine)
Three successive reactions can take place depending on the quantity and concentration of the reagents:
Nucleophilic attack of ethylamine (I) on iodomethane (II):
 The acid-base reaction (proton exchange) between ethylamine (I) and ethylmethylammonium ion releases ethylmethylamine( III )
The acid-base reaction (proton exchange) between ethylamine (I) and ethylmethylammonium ion releases ethylmethylamine( III )
Nucleophilic attack of ethylmethylamine (III) on iodomethane (II):
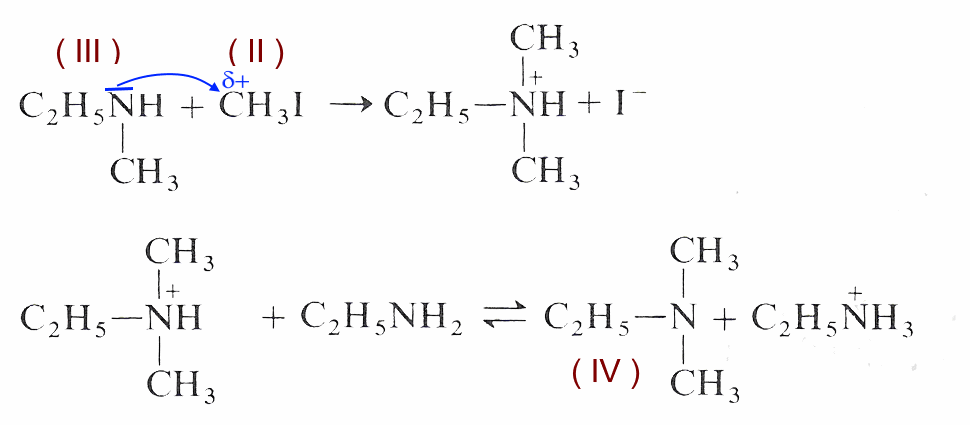 The acid-base reaction (proton exchange) between ethylamine (I) and diethylmethylammonium ion releases ethyldimethylamine (IV)
The acid-base reaction (proton exchange) between ethylamine (I) and diethylmethylammonium ion releases ethyldimethylamine (IV)
Nucleophilic attack of ethyldimethylamine (IV) on iodomethane (II):
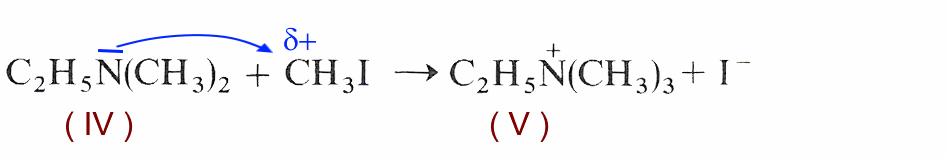 In the absence of mobile hydrogen on the ethyltrimethylammonium ion (V) formed, an acid-base reaction (proton exchange) can no longer take place.
In the absence of mobile hydrogen on the ethyltrimethylammonium ion (V) formed, an acid-base reaction (proton exchange) can no longer take place.