







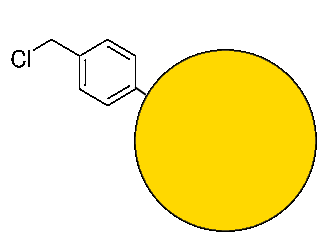
Anchor
A styrene-based copolymer to which a chloromethyl group is attached is the "anchor" by which the synthesis begins:
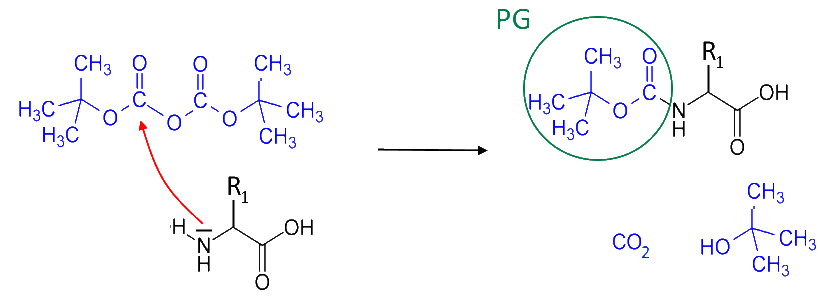
Protection of the first amino acid
We would like this amino acid not to react by its amine group $ -NH_2 $. Then it is protected by attaching a tertbutyloxycarbonyl group ( "PG ") by reaction with the corresponding acid anhydride:
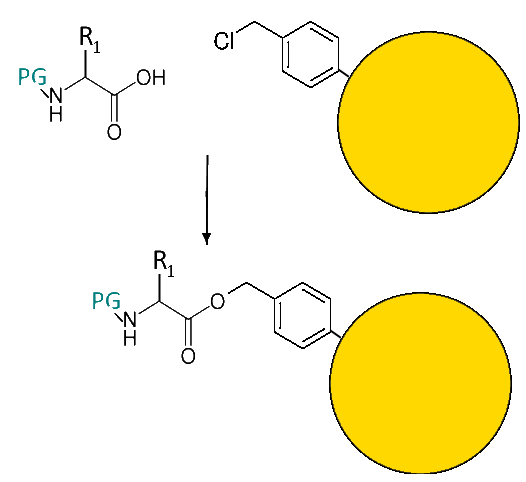
Fixing the first amino acid on the "anchor":
"The anchor" and the carboxyl group of the first amino acid are esterified. This is the only possible reaction, since the amine group is blocked!
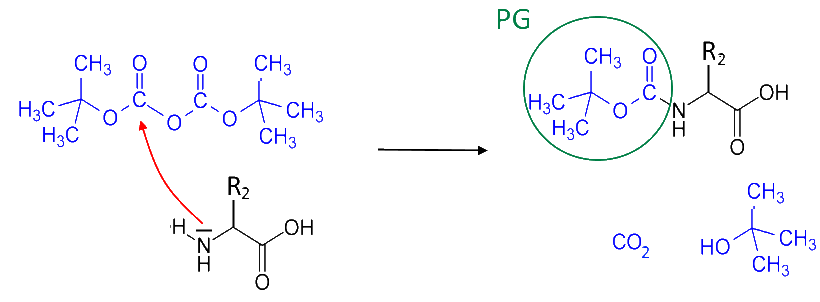
Protection of the second amino acid
A second amino acid sees its amine group protected :
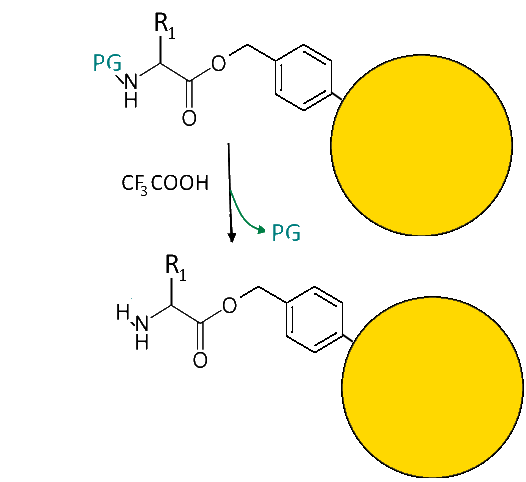
Deprotection of the first amino acid
The protective group is removed from the first amino acid by reaction with trifluoroacetic acid:
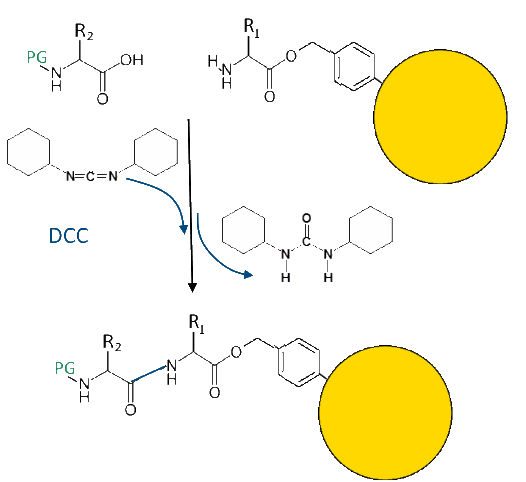
Peptide bond
Dicyclohexylcarbodiimide (DCC) establishes the peptide bond by fixing the eliminated water:
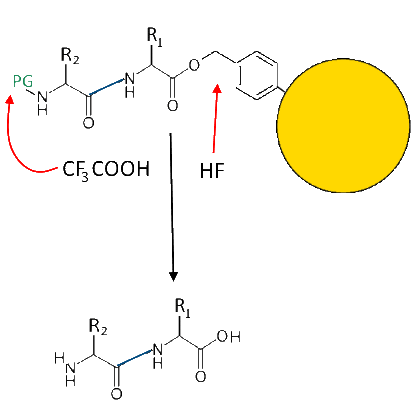
Peptide release
The protective group is eliminated as above, the hydrofluoric acid breaks the bond with "anchor" and here is the dipeptide!
This method was developed by:

Robert Bruce Merrifield (1921 - 2006)
It can be seen that the method can be used to fix as many amino acids as desired by repeating the synthesis before the final release of the desired peptide! The yield is excellent and several firms currently offer the production of a polypeptide in a few days!