Acid-base titration
Tutorial 1


 pH during titration of a strong base with a strong acid
Schematic:
pH during titration of a strong base with a strong acid
Schematic:
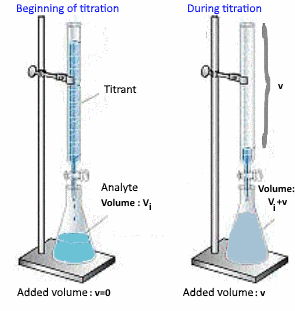
Determination of the volume of acid $ V_e $ added at the equivalent point
At E.P.:
$n_{acide\;added}= n_{base\;initial}$
$V_e\cdot c_{acid}=V_i\cdot c_{base}$
so $V_e$ is found.
Calculation of pH ( $ v $ volume of added acid)
$v=0$
pH of a strong base of molarity $c_{base}$:
$pH=14+logc_{base}$
$v\lt V_e$
Determine the numbers of base moles $ n_b $ that have not yet reacted, then:
$pH=14+log\frac{n_b}{V_i+v}$
$v\;=\;V_e$
$pH=7$
$v\gt V_e$
Determine the number of moles of acid $ n_a $ in excess
$pH=-log\frac{n_a}{V_i+v} $
$ 20 \; mL \; NaOH \; 0.05 \; M $ titrated by $ HCl \; 0.1 \; M $
Calculate the volume $ V_e $ of acid added at the equivalent point
For answers, use (possibly several times) the arrows ↑ Down! and ↓ Up!
Complete please this question before moving on to the next one!
$V_e\cdot c_{acid}=V_i\cdot c_{base}$
$V_e\cdot 0.1=0.020\cdot 0.05$
$V_e=0.010\;L$



