





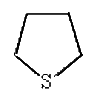
Thiolane
Name of the heterocycle: Prefix - Root(Body) - Suffix
Prefixes design the nature of heteroatoms, f.i.: Ox(a) = Oxygen Az(a) = Nitrogen Thi(a) = Sulfur In front of a vowel one leaves the "a" of coast
The root designates the number of ringmembers of the cycle: ir = 3 Ringmembers et = 4 Ringmembers ol = 5 Ringmembers = 6 Ringmembers (!) ep = 7 Ringmembers oc = 8 Ringmembers on = 9 Ringmembers ec = 10 Ringmembers
The suffix designates the degree of unsaturation. It depends on the number of ringmembers of the cycle and the nature of the heteroatom: Wholly Insaturated: 3 Ringmembers: ene (Exception for 1 N atom ): ine 4 Ringmembers: e 5 Ringmembers: e 6 Ringmembers: ine 7 Ringmembers: ine 8 Ringmembers: ine 9 Ringmembers: ine 10 Ringmembers:ine Saturated (at least one heteroatom N): 3 Ringmembers: idine 4 Ringmembers: idine 5 Ringmembers: idine 6 Ringmembers: ane 7 Ringmembers: ane 8 Ringmembers: ane 9 Ringmembers: ane 10 Ringmembers:ane Saturated (at least one heteroatom N): 3 Ringmembers: ane 4 Ringmembers: ane 5 Ringmembers: ane 6 Ringmembers: ane 7 Ringmembers: ane 8 Ringmembers: ane 9 Ringmembers: ane 10 Ringmembers:ane
For several prefixes, we have the priority: S > O > N
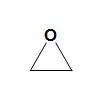 Oxirane
Oxirane
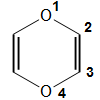 1,4-dioxine
1,4-dioxine
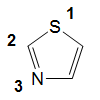 1,3-thiazole
1,3-thiazole
In cases of possible ambiguity, the position of the H - atoms attached to non-doubly bond atoms must be specified:
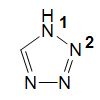 1H-Tetrazole
1H-Tetrazole
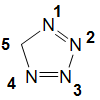 5H-Tetrazole
5H-Tetrazole
In cases of partial unsaturation, the nomenclature of the fully unsaturated cycle is retained while specifying the hydrogen atoms on the ring atoms where the double bond is missing: H - atoms attachments to non-doubly bound atoms:
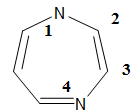 1H-1,4-Diazepine
is the basis of the name of
1H-1,4-Diazepine
is the basis of the name of
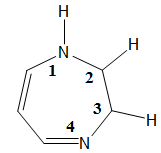 2,3-Dihydro-1H-1,4-diazepine
2,3-Dihydro-1H-1,4-diazepine