





Soap is a fatty acid salt which dissolves in water. Carboxylate ions have a long hydrocarbon chain that is relatively nonpolar, because the electronegativity difference between C and H is low. However their carboxylate group is ionic and strong differences of electronegativities make that it is polar:
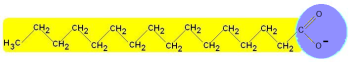
The carboxylate ion has an ionic hydrophilic head (water-loving, polar) and a long hydrophobic tail (water-fearing, non-polar)
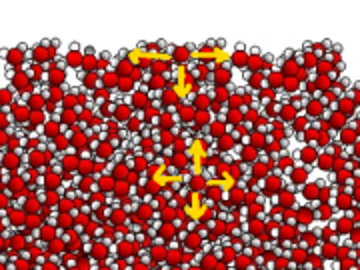
The water spider moves on the water because the water surface of the water is stretched as a membrane :

Due to the strong dipole-dipole attractions, the water molecules are packed in surface, creating a kind of membrane, whereas in the body of water these forces balance mutually.
The surface tension keeps coins on the water surface. Dissolved soap is added. Watch what happens:
Adapted from a video of → Berkeley Lab
The carboxylate ions are moving in the soap surface with their end directed toward the hydrophilic water. This results in a membrane formed by the hydrophobic tails of which the mutual attraction is very low. The surface tension is lowered. The gerri could not walk on a soapy water!

Soap is surfactant
You see skim milk under a microscope
The emulsion is formed by lipid (nonpolar) bubbles surrounded by carboxylate ions with their hydrophobic tails immersed in the lipid. As the hydrophilic heads repel by their negative charges, the emulsion remains stable.
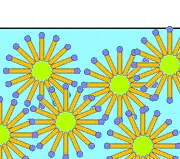
Soap is emulsifying
A greasy spot stains a cloth

Here's how the carboxylate ions of the soap take the spot off the fabric ready to be washed:
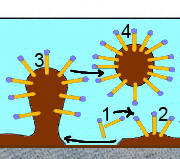
Soap is wetting
Soap bubbles have sometimes a surprisingly long life

This is why a double layer of carboxylate ions stabilize a thin layer of water with the hydrophilic heads facing the water!
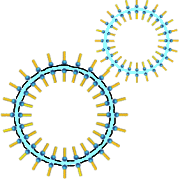
Soap is foaming