






By studying a salt of (2,3-dihydroxybutanedioic acid) tartaric acid under the microscope, Louis Pasteur noticed two different kinds of crystals:
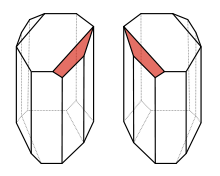
The superposition of the crystals is impossible, they are chiral objects
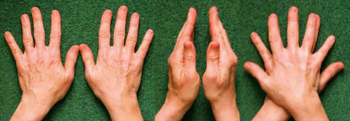
Image from → Vanderbilt University
Right hand and left hand are not identical, but images of each other in a mirror. This property is called chirality:

Chirality is common and applies for example to snails, screws, coil springs, crystals etc. ..
Pasteur recognized the cause of the chirality of the tartrate crystals within the tetrahedral carbon atom:
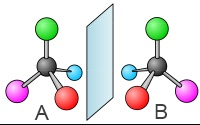
When an atom C has four different substituents, then this is a chiral object. They say it is asymetric. A and B are called enantiomers or optical isomers.
Two enantiomers - have 4 different substituents on the asymmetric carbon atom - are not identical - are images of one another in a mirror - have many of the same chemical properties - can only be distinguished in the presence of a reactive or phenomenon itself chiral

Comparison: These two screws are opposite chiral objects. A nut, chiral too, distinguishes them!