





Click 1, then 2 !

A nucleophile is a molecule or an anion which has a pair of unpaired electrons. An electrophilic center is a part of a molecule or a cation which attracts the electron pair of the nucleophile to enable it to form a dative bond.
Nucleophilicity depends on several factors: 1) Charge : The reagents that have one or more negative charges on the atom bearing the doublet are better than similar nucleophilic reagents with only a partial negative charge or any negative charge on the atom in question. That is why the negative charges repel their doublets and make them more accessible! By descending order of nucleophilicity we have, for example:

ethanolate

ethanol
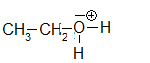
ethyloxonium
2) Electronegativity : The atoms that are more electronegative are less nucleophilic than similar atoms in the same period. That is why the more electronegative atoms retain their electron pairs stronger which are thus less available! By descending order of nucleophilicity we have, for example:

ethanamine

ethanol

chloroethane
We see that the nucleophilicity decreases from left to right in a period of the periodic table: N> O> Cl 3) Size of the atom supporting the doublet : The atoms that are larger are more nucleophilic than similar atoms in the same family. That is why the larger atoms retain their electron pairs (which are farther from the nucleus) less strongly ! By descending order of nucleophilicity we have, for example:

iodoethane

bromoethane

chloroethane
We see that the nucleophilicity increases from top to bottom in a family in the periodic table: Cl < Br < I 4) Inductive effect of groups attached to the atom bearing the doublet : Groups with I + effect increase the nucleophilicity. Indeed, they pump electrons to the atom bearing the doublet. These new electrons repel the doublet and thus make it more accessible. The opposite is true for groups with I- effect. By descending order of nucleophilicity we have, for example:

propanamine

ethanamine
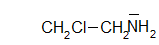
chloroethanamine
Propyl group exercises + I largest effect on the N atom as ethyl, chloroethyl group exerts a I- 5) Mesomeric effect of an aromatic system attached to the atom bearing the doublet It decreases the nucleophilicity. Indeed, the doublet is caused to participate in the mesomerie thereby decreasing the availability. By descending order of nucleophilicity we have, for example:

cyclohexanamine

aniline (phenylamine)
The phenyl group exercises an M- effect on the N atom.
Some examples from the site of Athénée de Luxembourg → here
:
