





A compound is a chemical substance whose molecules (or lattices) are made out of several kinds of atoms.
| Model | Formula | Name |
 | $H_2O$ | water |
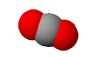 | $CO_2$ | carbon dioxyde |
 | $C_4H_{10}$ | butane |

The oxygen atom makes 2 bonds (it is bound in 2 "places"): it has valence 2. One hydrogen atom H (f.i. the right one) makes only one bond : it has valence 1.
Each atom has one main valence:

Often valences allow to foresee the formulas of compounds: Think about atoms being Lego blocks whose sizes would correspond to their valences and build a molecule like a straight wall:








Bigger molecules contain often the same atom groups (wrongly called "radicals") to which can be assigned valences, for instance for the hydroxide group OH :

| Nom | Groupe | Valence |
| Hydoxide | $OH$ | 1 |
| Nitrite | $NO_2$ | 1 |
| Nitrate | $NO_3$ | 1 |
| Carbonate | $CO_3$ | 2 |
| Sulfite | $SO_3$ | 2 |
| Sulfate | $SO_4$ | 2 |
| Phosphate | $PO_4$ | 3 |
| Ammonium | $NH_4$ | 1 |
Think now about groups being also Lego blocks whose sizes would correspond to their valences and use the method of the "straight wall" to find the formulas of their compounds, f.i.: $NaOH$ $NH_4I$ A$l(NO_2)_3$ $NH_4NO_3$ $BPO_4$ $H_2SO_4$ $(NH_4)_3PO_4$ A$l(OH)_3$ $(NH_4)_2S$ $H_2CO_3$ $HNO_3$ $Ca_2(PO_4)_3$
The name of compound formed by two atoms or groups X and Y has 3 different parts:
| Y | X | Suffixe |
| sodium | brom | ide |
| ammonium | chlor | ide |
| magnesium | sulf | ide |
| aluminium | carbon | ate |
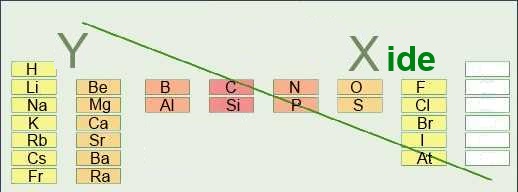
- X is the name of the atom situated higher and more to the right side in the periodic table. Its name suffix is often ide Special cases: O : Oxide ; S : sulfide; N: nitride; P: phosphide; C: carbide - Y is the normal name of the atom situated lower and more to the left side in the periodic table. - The groups, except ammonium, occupy the place of X in these names. Examples:
| $Ca$$O$ : | calcium oxide |
| $Mg$$I_2$ : | magnesium iodide |
| $NH_4$$Cl$ : | ammonium chloride |
| $Na_3$$PO_4$ : | sodium phosphate |
| $Sr$$(OH)_2$ : | strontium hydroxide |
| A$l_2$$(SO_3)_3$ : | aluminium sulfite |
The acids are compounds from hydrogen with nonmetals or groups Examples: $HCl$; $H_2SO_4$; $H_2CO_3$ Exceptions: $H_2O$: water ; $NH_3$: ammonia; $PH_3$: phosphine; $CH_4$: methane
Acids have special names according to the rule :
| hydrogen (..)ide | $\longrightarrow$ | (..)hydric acid |
| hydrogen (..)ite | $\longrightarrow$ | (..)ous acid |
| hydrogen (..)ate | $\longrightarrow$ | (..)ic acid |
for instance:
| hydrogen chloride | $\longrightarrow$ | chlorhydric acid |
| hydrogen sulfite | $\longrightarrow$ | sulfurous acid |
| hydrogen phoshate | $\longrightarrow$ | phosphoric acid |
Some little corrections are made for euphonious reasons !
The hydrogen salts contain hydrogenated groups whose names are for instance:
Groups or atoms with valence 2 :
| group+H | nom | valence |
| HS | hydrogen sulfide | 1 |
| HSO3 | hydrogen sulfite | 1 |
| HSO4 | hydrogen sulfate | 1 |
| HCO3 | hydrogen carbonate | 1 |
Groups or atoms with valence 3 :
| group+xH | nom | valence |
| HPO4 | hydrogen phosphate | 2 |
| H2PO4 | dihydrogen phosphate | 1 |
The addition of one H reduces the valence by one unit !
So we find the names of hydrogenated salts:
| Mg(HS)2 | magnesium hydrogen sulfide |
| LiHSO3 | lithium hydrogen sulfite |
| NaH2PO4 | sodium dihydrogen phosphate |
| Al2(HPO4)3 | aluminium hydrogen phosphate |