





An elementary substance is a chemical substance whose molecules (or lattices) are only formed out of one kind of atoms.
Metals are formed of lattices in their solid state, the atoms are simply piled up like marbles in a box. The fundamental structural unit is the single atom. Here are for instance formulas of metals:
| Copper: | Cu | Iron: | Fe | Mercury: | Hg |
- Metals have a particular metallic luster - Metals conduct electrical current - Metals conduct heat

Most nonmetallic elementary substances are made out of molecules. In the case of noble gases, the "molecules" are made out of one single atom ("monoatomic molecules"). In a few rare cases, the atoms are bound in such a complicated way that a molecular structure can no more be distinguished. (Red phosphorous, graphite carbon , diamond carbon)
- Nonmetals (except C) conduct electricity poorly
- Nonmetals conduct heat poorly

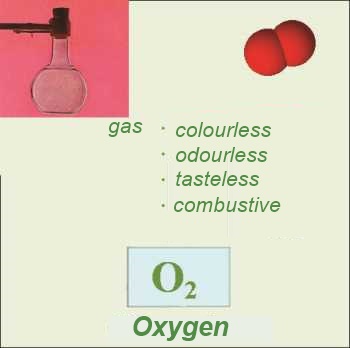




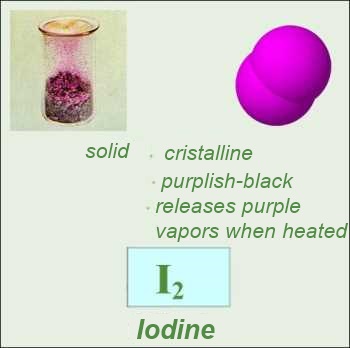 Formulas:
- The number of atoms in a molecule is written as a small number to the right of the symbol.
- The number of molecules is written before the formula.
Formulas:
- The number of atoms in a molecule is written as a small number to the right of the symbol.
- The number of molecules is written before the formula.
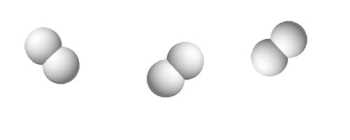
$3H_2$
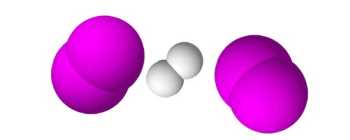
$H_2+2I_2$

$3S_8+2I_2$