






Svante Arrhenius (1859-927)
An acid is a substance which liberates in water the hydrogen ion $H^+$ A base is a substance which liberates in water the hydroxide ion OH^-
Chlorhydric acid $HCl$: $HCl$ $ _\longrightarrow^{water}$ $ H^+$ $ +$ $ Cl^-$ Sulfuric acid $H_2SO_4$: $H_2SO_4$ $ _\longrightarrow^{water}$ $ H^+$ $ +$ $ HSO_4^-$ Phosphoric acid $H_3PO_4$: $H_3PO_4$ $ _\longrightarrow^{water}$ $H^+$ $ +$ $ HPO_4^{2-}$ The base sodium hydroxide $NaOH$: $NaOH$ $ _\longrightarrow^{water}$ $Na^+$ $ +$ $ OH^-$ The base calcium hydroxide $Ca(OH)_2$: $Ca(OH)_2$ $ _\longrightarrow^{water}$ $Ca^{2+}$ $ +$ $ 2OH^-$
Colour indicators show whether a solution is acidic, basic or neutral, for instance bromothymol:
 in 1) acid 2) neutral 3) basic medium
Let's test this indicator with three salt solutions containing each two species of ions:
in 1) acid 2) neutral 3) basic medium
Let's test this indicator with three salt solutions containing each two species of ions:
| Ammonium chloride | Sodium chloride | Sodium hydroxide |
| $NH_4^+$ | $Na^+$ | $Na^+$ |
| $Cl^-$ | $Cl^-$ | $OH^-$ |
| yellow | green | blue |
It appears clearly that it is not the $Cl^-$ ion which is acidic, but rather the ammonium ion $NH_4^+$. Likewise it is not the $Na^+$ ion which is basic, but rather the $OH^-$ ion
The definition of acids and bases should not be limited to substances, but ions must also be included
Ions produced by the dissociation of an acid in water carry the electric current:
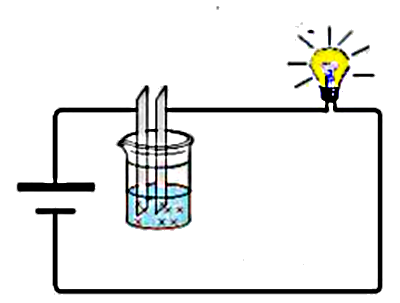
Let's test the influence of the solvent on that dissociation:
| Benzene | Alcohol | Water |
 |
 |
 |
It appears that alcohol allows also the dissociation of acids to ions!
The definition of acids and bases should not be limited to water as solvent.
The hydrogen atom $H$ has one proton making its nucleus and one electron making its electron cloud. If we take away this electron, we produce the hydrogen ion $H^+$ which is in fact nothing else than a mere proton. Because of its very small diameter and its positive charge, all surrounding molecules must be very strongly attracted by this ion, especially if they are polarized. ( the attraction between two charges of opposite signs is inversely proportional to the square of their distance !)!
The hydrogen ion $H^+$ cannot exist alone in an aqueous solution !
Therefore it was necessary to extend the simple Arrhenius definition to other chemical species , other solvents and to drop the notion of a free $H^+$ ion in solution. This important step was taken by Johannes Nikolas Brønstedt.