








Left: Copper blade in a vessel filled with a copper(II) sulfate solution Right: Zinc blade in a a vessel filled with a zinc sulfate solution 1 : electrical conductor 2: electrolytic junction filled with a saturated sodium sulfate solution A : ammeter
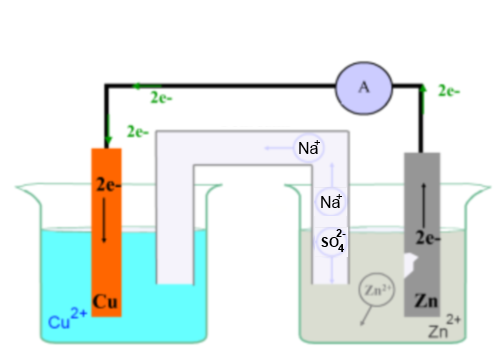
Zn looses electrons and is transformed to Zn2+ (colourless, enters into the solution ): oxydation The increase in positive charge is compensated by the migration of ions in the electrolytic junction

Cu2+ catches and is transformed to Cu (solid deposit on the copper blade) : reduction The loss of positive charge is compensated by the migration of ions in the electrolytic junction
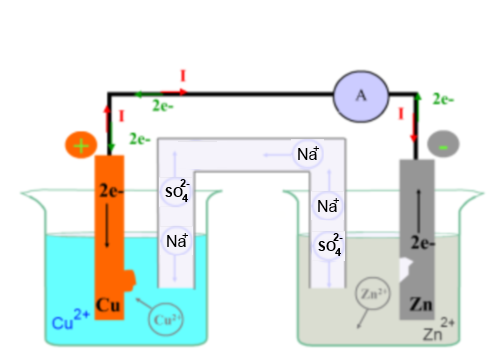
Electrons pass from the zinc blade to the copper blade The electrical current, as conventionally assumed, goes in the opposite direction The battery is a generator of electrical current ! Its positive + pole is, as conventionally assumed, where the current leaves, at the copper blade. Its negative - pole is, as conventionally assumed, where the current enters, at the zinc blade.